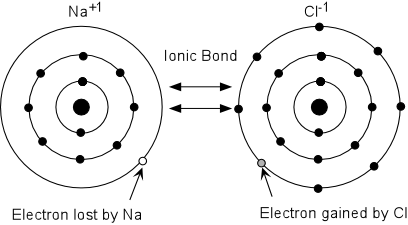
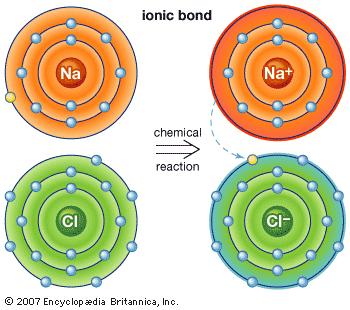
Ionic Bonds
Ionic Bonds is a bond in which one or more electrons from one atom are removed and attached to another atom, resulting in positive and negative ions which attract each other. These ions represent atoms that have lost one or more electrons and atoms that have gained one or more electrons. Ionic compounds conduct electricity when molten or in solution, but typically not as a solid.